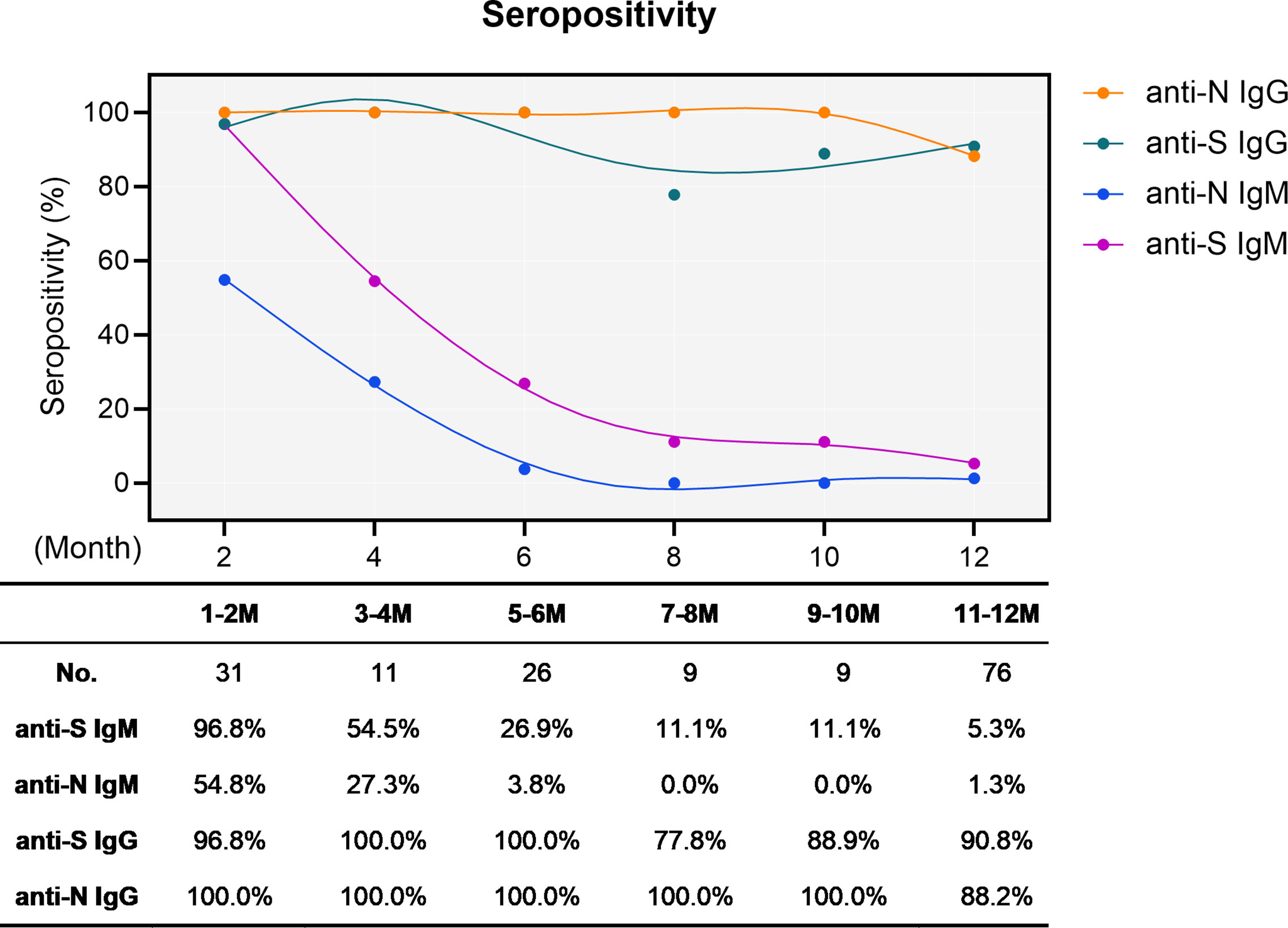
Blood samples were taken to obtain three time point antibody values: September 2020 (baseline, before vaccination), 14 (immunization peak) and 90 (long-term immunization not all volunteers, n = 27) days after the administration of the second dose of the vaccine. All patients were tested for SARS-CoV-2 nucleoprotein antibodies to ensure they had not been infected previously (Table 1 in S1 File). This observational study included 256 volunteer healthcare professionals vaccinated against SARS-CoV-2 (Comirnaty ™) at the Andújar Alto Guadalquivir Hospital who had not been previously infected by SARS-CoV-2. In addition, we will compare the results of two different testing platforms to describe the adjustments needed for each method for the appropriate measurement of the antibody load. The aim of this study is to describe the distribution of total S and IgG S antibodies in two cohorts -SARS-CoV-2 infected and non-infected- vaccinated volunteers-, and the titer evolution over time. Also, further evidence is needed on the distribution of the antibody titers in vaccinated volunteers for each vaccine and the evolution of the antibody titers over time, which is required to determine the need for booster doses in the near future. However, as most of the tests were intended for antibody testing in naturally infected volunteers, more data is needed for the use of the tests in vaccinated volunteers whose antibody levels are expected to be considerably higher, both if they have been previously infected or not. In all countries with ongoing vaccination plans at present, serological tests are now in the spotlight as a method to determine the efficacy of the vaccines. According to the Interim Guidelines for COVID-19 antibody testing from the CDC (Centers for Disease Control and Prevention), serological tests can play an important role in supporting the diagnosis of acute COVID-19 illness and, as a complement to qPCR, to increase the sensitivity of the nucleic acid detection method, but they should not be used to establish or exclude SARS-CoV-2 infection or reinfection, neither for the establishment of immune status nor until immunity is fully characterized. There are 200 serological techniques available worldwide to detect SARS-CoV-2 antibody isotypes (IgG, IgM, and IgA) as well as different combinations of them simultaneously. For most COVID-19 vaccines produced, this is the reason why the outcome priority was antibody generation by T cells, specifically neutralizing antibodies against spike protein containing RBD, the receptor binding domain. However antibodies to N are unlikely to neutralize the virus. Of the four structural proteins of the infectious virion, spike protein (S), N protein (nucleocapsid), M protein (matrix), and E protein (envelop), only N and S have been shown to trigger the production of high antibody titers in naturally infected patients. At the time this article was drafted, four vaccines had been approved in Europe by the European Medicines Agency: Comirnaty ™ (Pfizer-BioNtech), Vaxzebria (AstraZeneca), COVID-19 vaccine (Moderna) and COVID-19 vaccine (Janssen). There have been 626,985.198 cases of SARS-CoV-2 infection and 6,576.037 death around the world as of October 2022.Īs no effective treatment has been approved for human use during the ongoing pandemic, vaccines are the most powerful weapon against the SARS-CoV-2 pandemic to reduce the morbidity and mortality of COVID-19. On March 11, 2020, the World Health Organization declared the COVID-19 disease a pandemic, which is still a global threat two years and a half later. The novel SARS-CoV-2 was first described in the Chinese city of Wuhan in late 2019 and rapidly spread worldwide. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are within the paper and its Supporting information files.įunding: The author(s) received no specific funding for this work.Ĭompeting interests: The authors have declared that no competing interests exist.   
Received: AugAccepted: OctoPublished: November 3, 2022Ĭopyright: © 2022 Fernández-Suárez et al. PLoS ONE 17(11):Įditor: Robert Jeenchen Chen, Stanford University School of Medicine, UNITED STATES Citation: Fernández-Suárez A, Jiménez Coronado R, Clavijo Aroca C, Navarro Martín E, Qmega Qmega A, Díaz-Iglesias JM (2022) New insights into antibody levels against SARS-CoV-2 for healthcare personnel vaccinated with tozinameran (Comirnaty).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
